What Is The Makeup Of Clay Colloids In Alkaline Soils?
13 Soil Colloids
Colloids consist of clay minerals and organic matter, and play critical roles in soil chemic, physical, and biological backdrop. Colloids are very small-scale in size. The bulk of charges in soil comes from colloids, making them important for cation exchange capacity, buffering chapters, and retention of nutrients like Ca2+, K+, Mg2+, etc. In addition, the small size of colloids leads to very high surface areas, which facilitates chemical reactions and provides habitat for microbes. Some clays expand and contract with changes in soil wetness. The force from expanded clays can exist considerable, and commonly cause structural failures in house foundations and basement walls, roads and bridges, and other man-fabricated structures. Other clays do not expand, and make ideal substances for clay pottery, or tile roofs. The organic matter fraction of colloids are important for the development of soil structure, or as food sources for soil organisms. The soil properties attributed to colloids are numerous, and volition be the focus of the following laboratory activities.
- Measure the furnishings of unlike cations on colloidal backdrop.
- Identify the soil components controlling ion exchange.
- Determine the cation exchange capacity of selected soil horizons.
- Petri dishes
- Beakers
- Spatulas
- Pure kaolinite
- Pure bentonite
- Caliper
- 6V battery
- Solid copper wires, 2 total, 25 cm long
- Alligator clips for the wires
- Paper towels
- Test tubes
- Flasks
- Burettes
- Bottle-meridian dispensers gear up to dispense 10 mL
- Phenolphthalein indicator solution
- Aluminum chloride solution, 1 1000
- Potassium chloride solution, one M
- Sodium chloride solution, 1 M
- Calcium chloride solution, 1 Thousand
- Sodium hydroxide solution, 0.01 M
- Faux soil CEC excerpt solutions
- Norfolk E horizon, 0.002 M HCl
- 10 mL of excerpt represents ane g of soil
- Norfolk Bt horizon, 0.005 M HCl
- ten mL of excerpt represents 1 thou of soil
- Cecil Ap horizon, 0.010 M HCl
- ten mL of extract represents ane g of soil
- Cecil Bt Horizon, 0.007 M HCl
- x mL of excerpt represents 1 g of soil
- Whitestore Bt horizon, 0.015 M HCl
- ten mL of extract represents 0.5 g of soil
Using the recommended readings and the introduction to this lab, consider the questions listed below. These definitions/questions volition provide a concise summary of the major concepts to exist addressed in the lab. They will also serve equally the basis for the post-lab quiz and are useful study notes for exams.
- Define the term "soil colloid". What are the main types of soil colloids?
- Draw the sources of charges on soil colloids.
- Ascertain cation exchange chapters.
- What units are used to express cation substitution chapters?
- List the general quantity of cation exchange capacity contributed by kaolinite, montmorillonite (smectite), and humus.
- Define percentage base saturation. What cations are commonly considered base cations? Technically speaking they are non bases. Why are they chosen base cations?
Introduction
The extremely minor, colloidal particles (smaller than 0.001 mm) of dirt and humus control many of import chemical and physical properties of the soil. This portion of the soil is often called the "agile fraction". The small size of colloids results in a large surface area per unit weight, and their ionic structure results in a net electrical charge. The type, amount, and mineralogy of colloids will strongly influence most land management decisions. For example a soil that is 40% of clay that primarily consists of smectite (a 2:ane shrink-dandy clay) could have limitations for amalgam roads, or edifice foundations due to the shifting of the soil as the soil wets and dries. Such a soil could exist highly productive for row crop agriculture though, due to the loftier amount of charge that facilitates the retention of nutrients like Ca2+, K+, Mg2+, etc. On the contrary, a soil, such equally an Oxisol that has 80% clay has colloids that are primarily aluminum and iron oxides, which do non shrink or swell, and have a depression corporeality of charge. Thus, the soil would be well suited for building foundations. Notwithstanding, the high phosphorus fixation capacity, and the limited ability to retain base cations limit the productivity of the soil for row crop agriculture.
Ion exchange is one of the most significant features of the clay and humus fractions. The capacity of the particles to attract or adsorb cations is called the cation exchange chapters. This ability allows the soil to serve equally a storehouse of institute nutrients like potassium, calcium, and magnesium. This reactive substitution capacity also permits the soil to serve as a filter or treatment medium for land application of waste materials.
Determining Cation Exchange Capacity
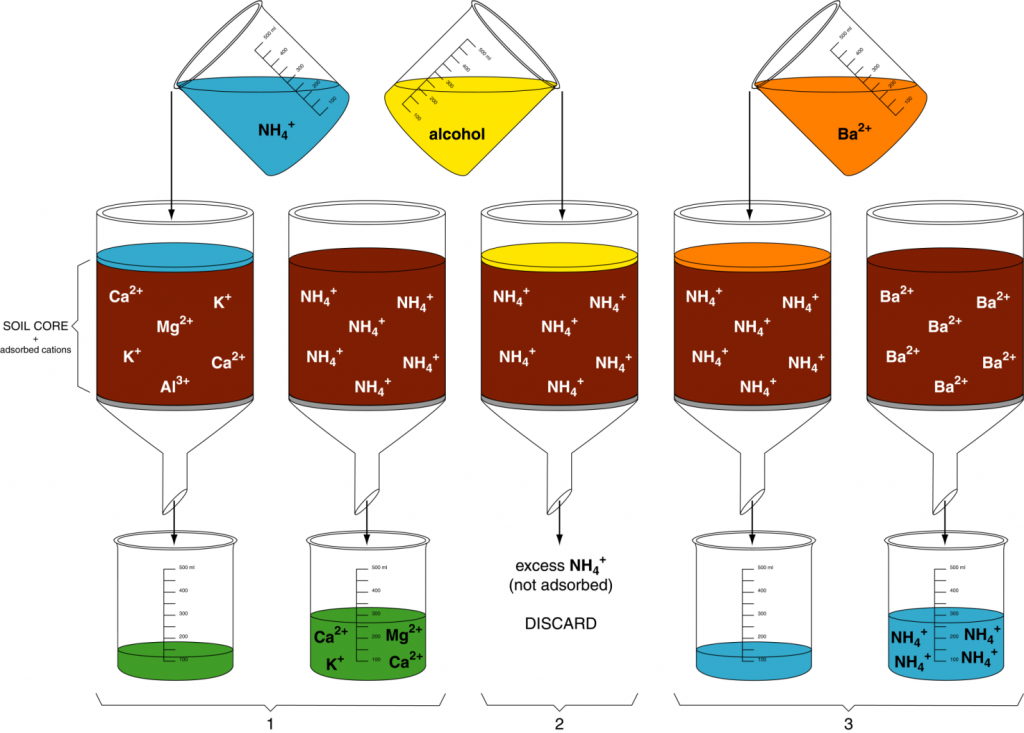
The cation exchange capacity (quantity of cations a soil can adsorb per unit weight, CEC) can be adamant using a simple displacement procedure (Figure xiii.i). In step 1, a soil sample is first saturated with a uncomplicated cation like NHiv + then all the negative charge sites are occupied by NHiv +. In step 2, excess NHiv + (i.east., not on exchange sites) is removed past leaching with ethyl booze. In pace three, another cation such every bit Ba2+ is used to readapt all the NH4 +. The NH4 + is nerveless in the filtrate and measured. The quantity of NH4 + collected from the sample is the quantity of cations that the soil can hold, i.due east. CEC.
Many cations could be used in step one of the displacement method. Soil laboratories often use the ammonium ion. Even so, in this activity, H+ will exist used as the saturating cation. Therefore, nosotros will determine the amount of extracted H+ in the filtrate. And then, using that amount, you volition calculate the CEC for each soil sample.
Calculating CEC uses the concept of moles of accuse. In chemistry, one mole of an element is the quantity of the element with a weight in grams numerically equal to its atomic weight. For example, the diminutive weight of K is 39.1, so ane mole of Thousand weighs 39.ane g. Ane mole of any element contains 6.02 x 1023 atoms of the element. Similarly, i mole of accuse is 6.02 x 1023 charges. If K exists every bit a cation in solution (One thousand+), then a solution containing a mole of Thou would contain 6.02 x 1023 positive charges.
The number of positive charges in the filtrate is thus equal to the number of negative charges on the exchange sites in the soil sample. Therefore, your task is to determine the number of charges in the filtrate. Each H+ ion has one positive charge, so past determining the amount of H+ in the filtrate, you tin can determine the number of positive charges in the filtrate and by extension, the number of negative charges on the commutation sites in the soil sample.
Activity 1: Sit-in of Shrink-Bully Characteristics
In a previous lab, you lot filled 2 petri dishes with slurries of bentonite and kaolinite and set them aside to dry. Remember those petri dishes and perform the following steps:
![]() Measure the height and radius of the discs of bentonite and kaolinite, and tape the results in Table 13.1.
Measure the height and radius of the discs of bentonite and kaolinite, and tape the results in Table 13.1.
![]() Summate the volume of each disc, and record the results in Table 13.1.
Summate the volume of each disc, and record the results in Table 13.1.
![]() Calculate and record the percent volume modify in each sample relative to the original volume (interior volume of the empty petri dish).
Calculate and record the percent volume modify in each sample relative to the original volume (interior volume of the empty petri dish).
Table 13.1. Shrink-Peachy Measurements & Volume Changes
| Sample | Radius (cm) | Tiptop (cm) | Volume (cm^iii) | % Volume Change |
|---|---|---|---|---|
| Empty petri dish | North/A | |||
| Kaolinite | ||||
| Bentonite |
![]() If yous were edifice a house with a basement, would you prefer the dominant clay mineral of the soil on your lot to be kaolinite or bentonite? Explain.
If yous were edifice a house with a basement, would you prefer the dominant clay mineral of the soil on your lot to be kaolinite or bentonite? Explain.
Activity two. Demonstrations of Colloid Charge
Using the remaining slurry from Activity ane, your instructor volition demonstrate colloid charge to the class. In this sit-in, two wires with alligator clips are attached to a 6V battery: one wire to the positive terminal, and the other to the negative terminal. The two wires are then inserted into the slurry and allowed to react for approximately xv minutes.
![]() Draw what y'all observe when the wires are removed from the slurry, and explain the phenomenon that causes this.
Draw what y'all observe when the wires are removed from the slurry, and explain the phenomenon that causes this.
Action 3: Demonstration of Flocculation and Dispersion
As y'all now know, most soil clays have a negative accuse that is neutralized to varying degrees by the adsorbed cations. Flocculates or aggregates form nearly rapidly and are nigh stable when the soil colloid is near completely neutralized by the adsorbed cation. The amount of neutralization is related to ion size, valence, and concentration. Perform the following experiment to exam the effect of various ions on the flocculation of a dispersed soil clay.
- Make full 5 exam tubes to ane/two full with the dispersed clay suspension. Test tube 5, containing the original dirt suspension, volition be reserved to utilize as a bank check.
- Add 10 drops of iM AlCl3 to test tube number one.
- Add x drops of 1 M KCl to test tube 2.
- Add together 10 drops of 1Grand NaCl to test tube 3.
- Add x drops of 1M CaClii to exam tube 4.
- Shake each suspension thoroughly.
- Observe and record the fourth dimension that flocculation starts in each tube.
- Go on to notice throughout the remainder of the class period and note the relative size and settling rates of the floccules.
![]() Record your observations in Tabular array 13.ii.
Record your observations in Tabular array 13.ii.
Table 13.2. Effect of cations on flocculation of a clay suspension.
| Added cation | Relative Size & Settling Rates of Floccules |
|---|---|
| M+ | |
| Na+ | |
| Ca2+ | |
| Al3+ | |
| Check |
Action 4. Determining CEC by replacing adsorbed cations.
In this activeness, you will titrate the filtrate with a 0.01 molar solution of NaOH using phenolphthalein as an indicator. Phenolphthalein changes from colorless to faint pink when the quantity of OH– ions added via the NaOH equals the quantity of H+ ions in the solution (that is, when the pH is raised to vii). For this activity, presume the soil samples take been extracted and the filtrates are now available for assay.
- Place 10 ml of each filtrate into separate 125 ml flasks. This 10 ml quantity is the amount of filtrate from one.0 gram of soil.
- Add 10 drops of the phenolphthalein indicator.
- Titrate the extract with the NaOH solution to a faint pinkish endpoint. The titration must exist done very advisedly to obtain meaningful results. If y'all put as well much NaOH in the flask and get a brilliant pink color, discard the solution and repeat the process. In the table beneath, record the milliliters of NaOH solution used to achieve the endpoint.
![]() Calculate the CEC and record your data in Table xiii.3.
Calculate the CEC and record your data in Table xiii.3.
Hither is an example of how to calculate the CEC, assuming ii.5 mL of NaOH was required to attain an end point. The reaction occurring during titration is
[latex]\text{NaOH}+\text{H}^{+}\rightarrow\text{Na}^{+}+\text{H}_2\text{O}[/latex]
Thus, ane mole of NaOH reacts with one mole of H+. Therefore, at the phenolphthalein end point, moles of NaOH added = moles of H+ in solution.
The solution of 0.01 molar NaOH contains 1 cmol charge per liter (1 cmolc/L). Therefore 2.5 mL NaOH contains
[latex]\text{cmol}_\text{c}\text{ of NaOH}=2.v\text{ mL NaOH}\times\frac{1\text{ L}}{thousand\text{ mL}}\times\frac{0.01\text{ mol NaOH}}{1\text{ 50}}\times\frac{1\text{ mol}_\text{c}}{i\text{ mol NaOH}}\times \frac{100\text{ cmol}_\text{c}}{1\text{ mol}_\text{c}}= 0.0025\text{ mol}_\text{c}\text{ NaOH}[/latex]
Thus, the CEC is
[latex]\frac{\text{cmol}_\text{c}}{\text{kg soil}}= \frac{0.0025\text{ cmol}_\text{c}}{1\text{ g soil}}\times\frac{1000\text{ yard soil}}{i\text{ kg soil}}= \frac{2.5cmolc}{\text{kg soil}}[/latex]
Table 13.three. CEC calculations.
| Extract | Molarity of NaOH | Milliliters NaOH used | Wt. of soil | CEC | % Clay |
|---|---|---|---|---|---|
| Norfolk E | 0.01 | one g | ix | ||
| Norfolk Bt | 0.01 | 1 chiliad | 37 | ||
| Cecil Ap | 0.01 | 1 g | 13 | ||
| Cecil Bt | 0.01 | i g | 51 | ||
| White Store Bt | 0.01 | 0.5 1000 | 57 |
![]() Using the data from Table 13.3, construct a graph in Figure 13.2 with percent clay on the X-axis and CEC on the Y-axis. Label the axes and plot all information. Draw a line through the points on the graph for the Norfolk Eastward, Norfolk Bt, and Cecil Bt, and answer the following questions:
Using the data from Table 13.3, construct a graph in Figure 13.2 with percent clay on the X-axis and CEC on the Y-axis. Label the axes and plot all information. Draw a line through the points on the graph for the Norfolk Eastward, Norfolk Bt, and Cecil Bt, and answer the following questions:
![]() Of the iv major factors that affect soil CEC (amount of clay, blazon of clay, amount of humus, pH), which is responsible for this linear increment in CEC?
Of the iv major factors that affect soil CEC (amount of clay, blazon of clay, amount of humus, pH), which is responsible for this linear increment in CEC?
![]() The slope of the line represents the alter in CEC divided by the modify in clay content. By expressing dirt content equally a fraction (for example, 30% clay = 0.30 kg clay/kg soil), the gradient becomes
The slope of the line represents the alter in CEC divided by the modify in clay content. By expressing dirt content equally a fraction (for example, 30% clay = 0.30 kg clay/kg soil), the gradient becomes
[latex]\text{Gradient}=\frac{\Delta\frac{\text{ cmol}_\text{c}}{\text{kg soil}}}{\Delta\frac{\text{kg dirt}}{\text{kg soil}}}=\frac{\text{max CEC}-\text{min CEC}}{\text{max clay}-\text{min clay}}=\text{______}\frac{\text{ cmol}_\text{c}}{\text{kg clay}}[/latex]
![]() The calculated gradient is the CEC on a clay basis. Using that slope (cmolc/kg clay), utilise Effigy xiii.ii to determine the type of clay in the Norfolk and Cecil soils.
The calculated gradient is the CEC on a clay basis. Using that slope (cmolc/kg clay), utilise Effigy xiii.ii to determine the type of clay in the Norfolk and Cecil soils.
![]() The line you have drawn does non pass through the origin, indicating that a soil with no clay would yet have CEC. How could this exist possible?
The line you have drawn does non pass through the origin, indicating that a soil with no clay would yet have CEC. How could this exist possible?
![]() The Cecil Ap horizon sample falls just above the line you drew in question #1, however both Cecil samples were taken from the same profile. How can you explain this difference in CEC?
The Cecil Ap horizon sample falls just above the line you drew in question #1, however both Cecil samples were taken from the same profile. How can you explain this difference in CEC?
![]() The CEC for the White Shop Bt horizon does not fall on the line for the Norfolk and Cecil soil either. Explain what may exist dissimilar about the colloids in the White Store Bt soil.
The CEC for the White Shop Bt horizon does not fall on the line for the Norfolk and Cecil soil either. Explain what may exist dissimilar about the colloids in the White Store Bt soil.
Example Calculations of Cation Substitution Chapters
Assume a soil has a CEC of one.0 cmolc/kg. How many kg of Ca2+ tin be adsorbed in a hectare of soil to a depth of 20 cm? Assume a bulk density of i.four Mg/g3.
Soil volume = expanse depth:
[latex]\text{Soil book}=10,000\text{ k}^ii\times0.ii\text{ thousand}=2,000\text{ m}^3[/latex]
Soil weight = density book:
[latex]\text{Soil weight}=1.iv\frac{\text{Mg}}{\text{k}^3}×2,000\text{ yard}^3=two,800\text{ Mg}=ii,800,000\text{ kg}[/latex]
Because Ca has a valence of 2, i mole of Ca has 2 moles of charge:
[latex]\frac{2\text{ mol}_\text{c}}{ane\text{ mol Ca}|^{2+}}[/latex] or in terms of cmols, [latex]\frac{ii\text{ cmol}_\text{c}}{i\text{ cmol Ca}|^{2+}}[/latex]
The soil tin can adsorb 1.0 cmolc/kg. The corporeality of Ca required to supply this corporeality of charge is
[latex]\frac{1\text{ cmol}_\text{c}}{\text{kg soil}}\times\frac{1\text{ cmol Ca}^{2+}}{2\text{ cmol}_\text{c}}=\frac{0.five\text{ cmol Ca}^{2+}}{\text{kg soil}}[/latex]
The atomic weight of Ca is twoscore.078, so the soil tin can adsorb
[latex]\frac{0.five\text{ cmol Ca}^{2+}}{\text{kg soil}}\times\frac{1\text{ mol Ca}^{two+}}{100\text{ cmol Ca}^{2+}}\times\frac{40.078\text{g Ca}^{2+}}{1\text{mol Ca}^{ii+}}=\frac{0.twenty\text{ thou Ca}^{2+}}{\text{kg soil}}[/latex]
The quantity of Ca adsorbed in the full soil volume is
[latex]\frac{0.20\text{ thousand Ca}^{2+}}{\text{ kg soil}}\times\frac{2,800,000\text{ kg soil}}{ane\text{ ha}}\times\frac{1\times\text{ kg Ca}^{2+}}{one thousand\text{ g Ca}^{ii+}}=\frac{560\text{ kg Ca}^{ii+}}{ha}[/latex]
In the problem to a higher place, how much Al3+ could be adsorbed past the soil?
Because the valence of Al is three, one mole of Al has 3 moles of charge. Thus, the soil, which has 1 cmolc/kg, can adsorb
[latex]\frac{1\text{ cmol}_\text{c}}{\text{kg soil}}× \frac{1\text{cmol Al}^{3+}}{3\text{ cmol}_\text{c}}=\frac{0.33\text{ cmol Al}^{three+}}{\text{kg soil}}[/latex]
The atomic weight of Al is 26.981538 and then the soil tin adsorb
[latex]\frac{0.33\text{ cmol Al}^{3+}}{\text{kg soil}}\times\frac{1\text{ mol Al}^{3+}}{100\text{ cmol Al}^{3+}}\times\frac{26.982\text{ g Al}^{3+}}{ane\text{ mol Al}^{3+}}=\frac{0.09\text{ g Al}^{three+}}{\text{kg soil}}[/latex]
The quantity of Al adsorbed in the total soil book is
[latex]\frac{0.09\text{ 1000 Al}^{3+}}{\text{ kg soil}}\times\frac{two,800,000\text{ kg soil}}{1\text{ ha}}\times\frac{1\times\text{ kg Al}^{iii+}}{k\text{ thousand Al}^{iii+}}=\frac{252\text{ kg Al}^{three+}}{ha}[/latex]
Cation Substitution Capacity Calculations
Calculate how many kg/ha of the following cations this soil (with a CEC of 1 cmolc/kg) could adsorb.
![]() Mg2+, with an atomic weight of 24.305:
Mg2+, with an atomic weight of 24.305:
![]() Na+, with an atomic weight of 22.990:
Na+, with an atomic weight of 22.990:
![]() H+, with an diminutive weight of 1.008:
H+, with an diminutive weight of 1.008:
![]() K+, with an atomic weight of 39.098:
K+, with an atomic weight of 39.098:
Base Saturation Calculations
Catwo+, Mg2+, K+, and Na+ are called basic cations because when they are in solution, the solution is basic. In contrast, H+ and Al3+ are considered acidic cations because they lower solution pH.
Therefore, base of operations saturation is
[latex]\text{Percentage base saturation}=\frac{\text{ cmol}_\text{c}\text{ of base of operations cations}}{\text{total CEC of the soil}}\times100\text{%}[/latex]
An alkaline soil constitute in a semi-arid region might have the following cation commutation characteristics (per kg of soil):
- ix.0 cmolc Ca2+
- 2.v cmolc Mg2+
- 0.5 cmolc M+
- two.0 cmolc Althree+
- 2.0 cmolc H+
![]() If these cations are the only ones on the exchange sites of this soil, what is the percent base saturation?
If these cations are the only ones on the exchange sites of this soil, what is the percent base saturation?
Activity 5. Calculating versus estimating CEC
There are two ways you tin can summate the CEC: the sum of cations method and the mineralogy method.
The Sum-of-Cations Method
If you have a soil analysis where the quantities of all cations in the soil are listed, just summing all those exchangeable quantities will yield the CEC yous plant in the preceding problems.
The "Mineralogy" Method
As y'all know from your reading and form discussion, clay minerals have a range of values for CEC. If the mineralogy of the dirt fraction is known (that is, the type and amounts of each clay mineral), then the CEC can be approximated.
To brand these calculations easier, Tabular array 13.4 contains representative values for CEC to use in all calculations for this class unless otherwise noted. In nature, however, these soil colloids will have a range of values.
Table 13.iv. Typical CEC of various soil colloids.
| Mineral or colloid type | CEC of pure colloid |
|---|---|
| cmolc/kg | |
| kaolinite | 10 |
| illite | 30 |
| montmorillonite/smectite | 100 |
| vermiculite | 150 |
| humus | 200 |
Every bit an example of this mineralogy approach to CEC calculations, consider a soil having 100% clay where the clay is 100% kaolinite. The CEC would then be 10 cmolc/kg. If a soil contains only 10% kaolinite (or 10 kg clay in 100 kg soil), however, this dirt would contribute
[latex]\text{Total CEC of the soil}=\frac{10\text{ cmol}_\text{c}}{\text{kg dirt}}\times\frac{10\text{ kg clay}}{100\text{ kg soil}}=\frac{1.0\text{ cmol}_\text{c}}{\text{kg soil}}[/latex]
A prairie soil contains 30% clay. This clay sized fraction is dominantly montmorillonite. The soil besides contains 5% humus (organic matter).
![]() Using the mineralogy method, what is the cation exchange capacity (CEC) contributed by the clay?
Using the mineralogy method, what is the cation exchange capacity (CEC) contributed by the clay?
![]() What is the estimated cation exchange capacity (CEC) contributed by the humus?
What is the estimated cation exchange capacity (CEC) contributed by the humus?
![]() What is the full estimated CEC of this soil?
What is the full estimated CEC of this soil?
The post-obit is actual laboratory data for the soil in the example:
Table 13.5. Exchangeable cation data
| Exchangeable Acerbity | Exchangeable Bases | Exchangeable Bases | Exchangeable Bases | Exchangeable Bases |
|---|---|---|---|---|
| H+ + Al3+ | Ca2+ | Mg2+ | 1000+ | Na+ |
| ----cmolc/kg---- | ----cmolc/kg---- | ----cmolc/kg---- | ----cmolc/kg---- | ----cmolc/kg---- |
| 14.0 | 29 | 10 | five.5 | one.5 |
![]() Calculate the CEC from the data in the table using the sum of cations method.
Calculate the CEC from the data in the table using the sum of cations method.
![]() What is the pct base saturation?
What is the pct base saturation?
![]() If you wanted to replace the Na+ with Catwo+, how many kilograms of Catwo+ per ii,800,000 kg of soil would you demand?
If you wanted to replace the Na+ with Catwo+, how many kilograms of Catwo+ per ii,800,000 kg of soil would you demand?
A quiz for this lab will exist available online. Delight admission it equally directed by your instructor.
Source: https://kstatelibraries.pressbooks.pub/soilslabmanual/chapter/soil-colloids/
Posted by: langstonhatepeor.blogspot.com

0 Response to "What Is The Makeup Of Clay Colloids In Alkaline Soils?"
Post a Comment